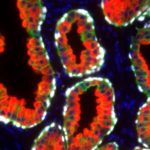
Anti-PSA (Prostate specific antigen) clone HAM18 stands out among more than 1,000 commercially available PSA antibodies because of its documented high specificity and sensitivity for recognition of prostatic cancer. Although PSA is solely expressed in prostatic epithelial cells, studies utilizing various different other antibodies have described PSA expression in 9%-60% of breast cancers 1-8, in 55% of malignant melanomas 7, 3%-9% of male breast cancers 8, 9, 3 of 3 acinar pancreatic cancers 10, various salivary gland carcinomas (including 6 of 11 pleomorphic adenomas, 1 of 6 mucoepidermoid carcinomas, and 1 of 2 adenocarcinomas not otherwise specified) 11-13. Multiple case reports have also described PSA expression in at least 4 cases of paraurethral adenocarcinoma 14-18 and in a urinary bladder cancer 19. Moreover PSA expression was described to occur in normal salivary glands, 60% of salivary gland adenomas, and in normal pancreatic epithelium 12. Using Anti-PSA clone HAM18, PSA positivity was not seen in any of these normal and neoplastic tissues.
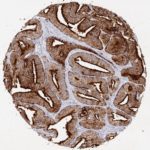
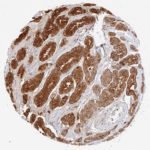
The validation of Anti-PSA clone HAM18 on more than 20,000 cancers using the protocol recommended by Oncodianova at an antibody dilution of 1:800 enabled a precise documentation of the sensitivity and specificity of this antibody. The key figures on the performance of the HAM18 Anti-PSA antibody are as follows.
Sensitivity
| Prostate Cancer Subset | Samples analyzed (n) | PSA positive (n) | Sensitivity at 1:800 (1:100) dilution |
| Gleason 3+3=6 | 2,439 | 2,437 | 99.92% |
| Gleason 3+4=7 | 7,470 | 7,461 | 99.87% |
| Gleason 4+3=7 | 2,213 | 2,205 | 99.64% |
| Gleason 8 | 112 | 111 | 99.15% |
| Gleason 9-10: | 628 | 618 | 98.41% |
| Recurrent prostate cancer after therapy (Gleason ≥8) | 392 | 387 | 99.72% |
| Small cell carcinoma of the prostate | 13 | 1 | 7.69% |
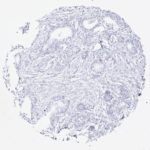
Specificity
| Tumor type | Samples analyzed (n) | PSA positive (n) | Specificity at 1:800 dilution |
| Adenocarcinoma,
extraprostatic |
877 | 1* | 99.89% |
| Squamous cell carcinoma, extraprostatic | 319 | 0 | 100% |
| Small cell carcinoma,
extraprostatic |
46 | 0 | 100% |
| Other extraprostatic carcinomas | 1,603 | 0 | 100% |
| All extraprostatic
malignancies |
2,845 | 1 | 99.96% |
*Weak extraprostatic PSA positivity was seen in 1 of 30 endometroid ovarian cancers
The use of highly validated antibodies is highly recommended for certified and accredited pathology laboratories.
In diagnostic routine, PSA (HAM18) immunohistochemistry can be used in the following applications:
- Carcinoma of unknown origin: Proof or rule out origin from prostatic cancer.
- Bladder tumor of male patients without unequivocal urothelial precursor lesion suggesting urothelial origin: Rule out origin from a prostate cancer.
- Advanced high-grade prostate cancer with rather low (or unknown) serum PSA levels: Low PSA expression in poorly differentiated cancer suggest that serum PSA levels may “underestimate” total tumor mass of the patient.
Reference for clone HAM18:
Bonk S et al. Prognostic and diagnostic role of PSA immunohistochemistry: A tissue microarray study on 21,000 normal and cancerous tissues. Oncotarget 2019 (10): 5439-5453. https://doi.org/10.18632/oncotarget.27145
Further References
[1] Poh BH, Jayaram G, Sthaneshwar P, Yip CH: Prostate-specific antigen in breast disease. Malays J Pathol 2008, 30:43-51.
[2] Alanen KA, Kuopio T, Collan YU, Kronqvist P, Juntti L, Nevalainen TJ: Immunohistochemical labelling for prostate-specific antigen in breast carcinomas. Breast Cancer Res Treat 1999, 56:169-76.
[3] Heyl W, Wolff JM, Biesterfeld S, Schroder W, Zitzelsberger D, Jakse G, Rath W: Immunohistochemical analysis of prostate-specific antigen does not correlate to other prognostic factors in breast cancer. Anticancer Res 1999, 19:2563-5.
[4] Miller MK, Unger PD, Bleiweiss IJ: Immunohistochemical analysis of prostate specific antigen in breast cancer. Breast Cancer Res Treat 2001, 68:111-6.
[5] Ilvan S, Celik V, Cetinaslan I, Calay Z, Ferahman M: Immunohistochemical analysis of prostate-specific antigen in female breast cancer. J BUON 2004, 9:183-6.
[6] Narita D, Cimpean AM, Anghel A, Raica M: Prostate-specific antigen value as a marker in breast cancer. Neoplasma 2006, 53:161-7.
[7] Bodey B, Bodey B, Jr., Kaiser HE: Immunocytochemical detection of prostate specific antigen expression in human primary and metastatic melanomas. Anticancer Res 1997, 17:2343-6.
[8] Kraus TS, Cohen C, Siddiqui MT: Prostate-specific antigen and hormone receptor expression in male and female breast carcinoma. Diagn Pathol 2010, 5:63.
[9] Carder PJ, Speirs V, Ramsdale J, Lansdown MR: Expression of prostate specific antigen in male breast cancer. J Clin Pathol 2005, 58:69-71.
[10] Kuopio T, Ekfors TO, Nikkanen V, Nevalainen TJ: Acinar cell carcinoma of the pancreas. Report of three cases. APMIS 1995, 103:69-78.
[11] van Krieken JH: Prostate marker immunoreactivity in salivary gland neoplasms. A rare pitfall in immunohistochemistry. Am J Surg Pathol 1993, 17:410-4.
[12] Elgamal AA, Ectors NL, Sunardhi-Widyaputra S, Van Poppel HP, Van Damme BJ, Baert LV: Detection of prostate specific antigen in pancreas and salivary glands: a potential impact on prostate cancer overestimation. J Urol 1996, 156:464-8.
[13] James GK, Pudek M, Berean KW, Diamandis EP, Archibald BL: Salivary duct carcinoma secreting prostate-specific antigen. Am J Clin Pathol 1996, 106:242-7.
[14] Svanholm H, Andersen OP, Rohl H: Tumour of female paraurethral duct. Immunohistochemical similarity with prostatic carcinoma. Virchows Arch A Pathol Anat Histopathol 1987, 411:395-8.
[15] Spencer JR, Brodin AG, Ignatoff JM: Clear cell adenocarcinoma of the urethra: evidence for origin within paraurethral ducts. J Urol 1990, 143:122-5.
[16] Zaviacic M, Sidlo J, Borovsky M: Prostate specific antigen and prostate specific acid phosphatase in adenocarcinoma of Skene’s paraurethral glands and ducts. Virchows Arch A Pathol Anat Histopathol 1993, 423:503-5.
[17] Ebisuno S, Miyai M, Nagareda T: Clear cell adenocarcinoma of the female urethra showing positive staining with antibodies to prostate-specific antigen and prostatic acid phosphatase. Urology 1995, 45:682-5.
[18] Sloboda J, Zaviacic M, Jakubovsky J, Hammar E, Johnsen J: Metastasizing adenocarcinoma of the female prostate (Skene’s paraurethral glands). Histological and immunohistochemical prostate markers studies and first ultrastructural observation. Pathol Res Pract 1998, 194:129-36.
[19] Grignon DJ, Ro JY, Ayala AG, Johnson DE, Ordonez NG: Primary adenocarcinoma of the urinary bladder. A clinicopathologic analysis of 72 cases. Cancer 1991, 67:2165-72.