learn
ABOUT our work
-
ONCOdianova is an antibody manufacturing network and concentrates on the tissue-based detection of cancer immunology checkpoint biomarkers.
-
Our vision is to provide the best primary antibodies for combined use in multiplex IHC panels.
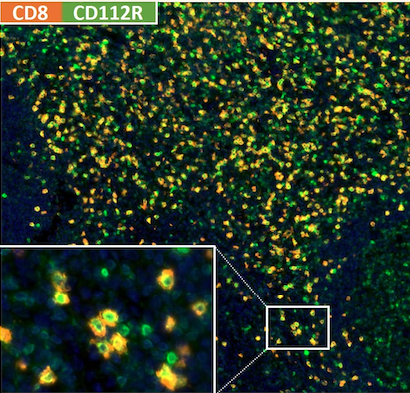
Multiplex immunohistochemistry will allow to identify what cell expresses the target, not just whether the target is present. The complexities of immune checkpoint pathways along with the interplay of the tumor cells, the microenvironment and systemic immune parameters offer opportunities for research and clinical translation.
We believe that IHC-assessment of predictive immune checkpoint targets in tumor tissues will be crucial and indispensable to select patients and differentiating drugs to optimal guide treatment by immunotherapy.
read our