Specific references from the American Association of Cancer Research (AACR) annual meeting 2020
Proceedings: AACR Annual Meeting 2020; April 27-28, 2020 and June 22-24, 2020; Philadelphia, PA, Volume 80, Issue 16 Supplement, pp. 3870 and pp. 4970
Abstract 3870:
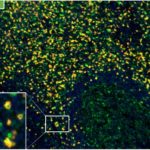
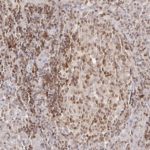
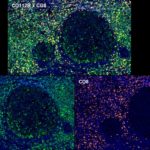
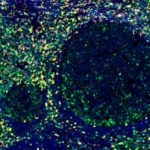
Patterns of CD112R expression in normal lymphatic tissues, inflammation and cancer
DOI https://doi.org/10.1158/1538-7445.AM2020-3870
Abstract 4970:
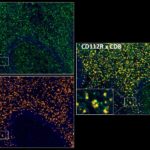
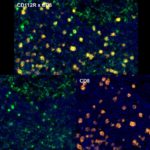
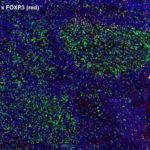
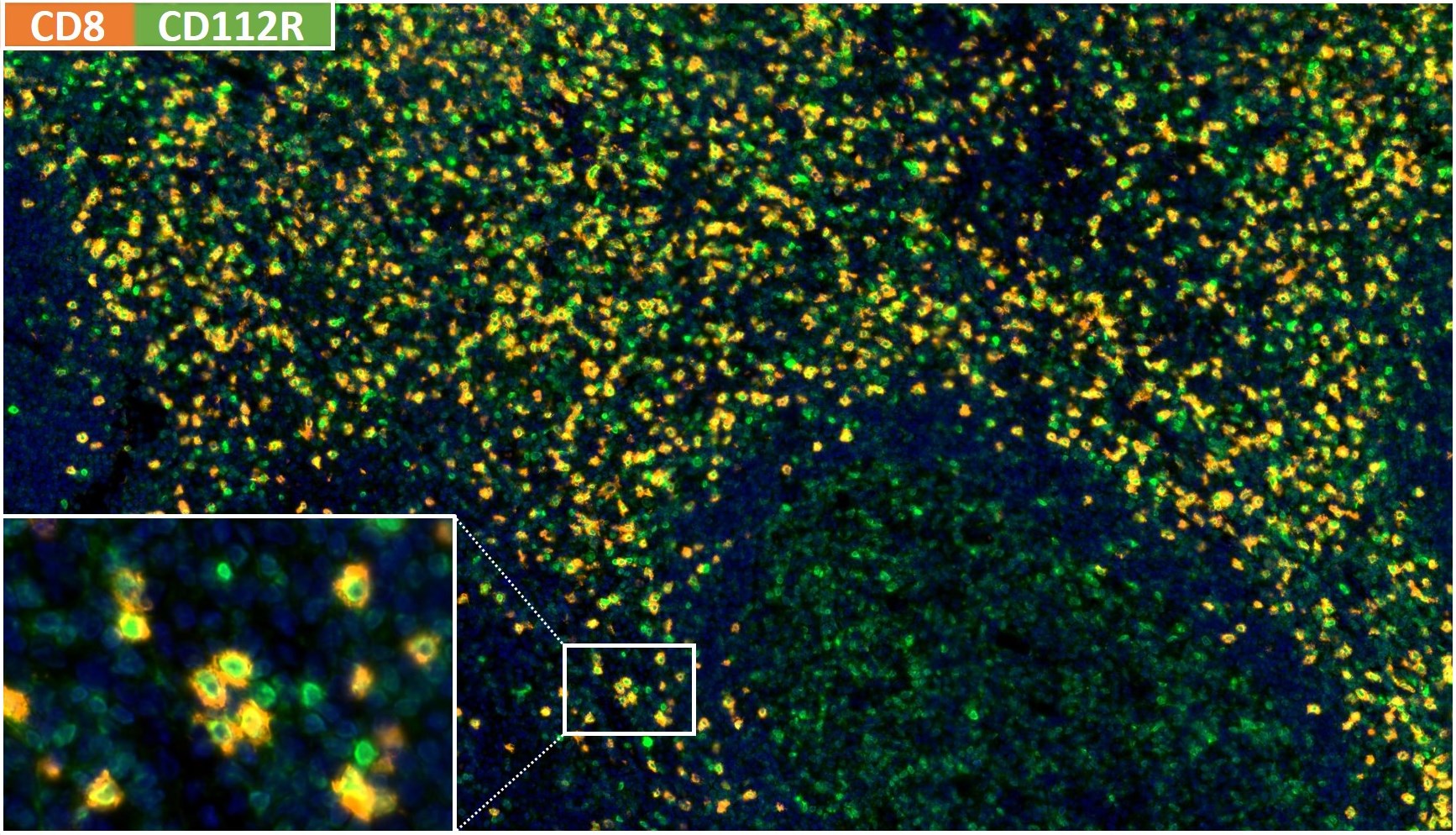
Prognostic role of CD112R, PD-1 and Ki67 expression in CD8+cytotoxic T cells in colorectal cancer
DOI https://doi.org/10.1158/1538-7445.AM2020-4970
Anti-PVRIG/CD112R clone R12 has been developed for detection of CD112R in routine formalin-fixed paraffin-embedded tissue specimen (IHC FFPE) to be used in bright field immunohistochemistry and moreover for multicolor immunofluorescence (fluorescence multiplex IHC). Anti-PVRIG/CD112R clone R12 has been validated for sensitivity and specificity on large quantities of normal and tumor tissues.
Clone R12 displays no background in non lymphoid cells and in epithelial cells. Based on the high specificity combined with an inherent high signal to noise ratio clone R12 is ideally suited for multiplexed immunohistochemistry studies of CD112R in human tissue specimen.
Initial clinical studies for CTLA4 and PD-1 inhibitors have shown that the blocking of immune checkpoint ligand-receptor interactions can supports the immune systems to fight tumor cells. The immune checkpoint axis TIGIT-PVR/PVRL2 has become a promising target in cancer immunotheraphy. T cell immunoglobulin and ITIM domain (TIGIT) and CD226 constitute a T cell cosignaling pathway in which CD226 and TIGIT serve as costimulatory and coinhibitory receptors for the ligands CD155 and CD112, respectively. Moreover, this TIGIT signaling axis includes a more complex receptor ligand system with markers like CD112R, CD96 and DNAM-1, which all serve as potential targets for immunotherapeutic interventions.
CD112R, a member of poliovirus receptor-like proteins, is preferentially expressed on T-cells and inhibits T-cell receptor mediated signals. CD112R binds to its ligand CD112 which is widely expressed on antigen-presenting cells and on tumor cells. CD112R competes with CD226 to bind to CD112 and thereby acts as a coinhibitory receptor for T cells. Blockage of the CD112R–CD112 interaction enhances human T cell response.
Poliovirus receptor (PVR)-like proteins are originally known to mediate epithelial cell-cell contacts and have shown T cell cosignaling functions. The two ligands, CD112 (PVRL2/nectin-2) and CD155 (PVR/Necl-5), interact with CD226 (DNAM-1) to costimulate T cells. Interaction with another coinhibitory receptor, TIGIT (T cell Ig and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain) inhibits T cell response. The interaction of TIGIT and CD112 is very weak and CD155 seems to be the major ligand in this complex network. Moreover, CD96 (a PVR-like protein present on T cells and NK cells) interacts with CD155 but not with CD112.
Basic and clinical research on relationships in the complex ligand/receptor network for CD112, including CD112R, CD226, and TIGIT will provide a better understanding for immunotherapheutic approaches.